Triple receptor agonist · GLP-1, GIP, and glucagon
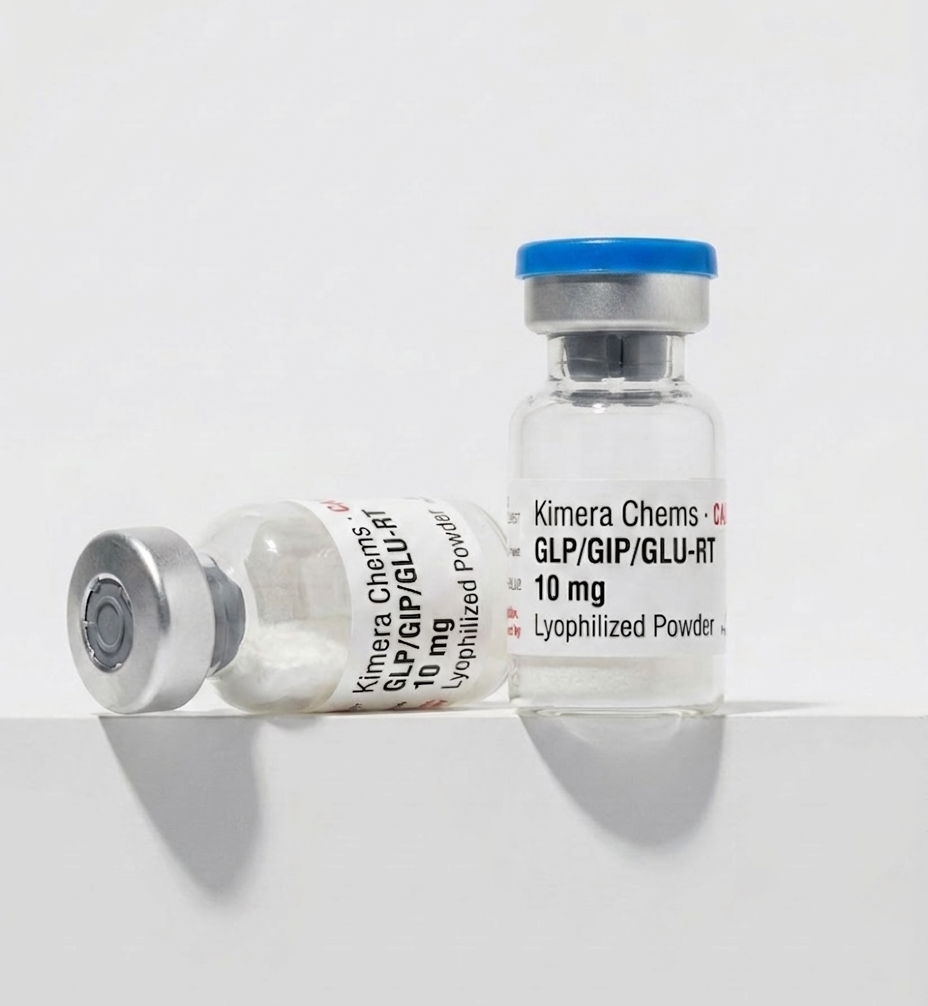
Retatrutide (LY3437943) · 10mg
Investigational triple-agonist peptide developed by Eli Lilly. The most-studied next-generation candidate in the GLP-1 class, with Phase 2 data showing dose-dependent body weight reduction. Supplied as lyophilized powder, >99% HPLC purity.
Select Pack
Quantity
Total
17,995EGP
Cash on delivery or instant transfer · Order confirmed on WhatsApp
Current Batch · Independently Verified
Third-party tested by an accredited US analytical lab. Full COA available on request.
- Verified Purity
- 99.637%
- Assay
- 11.179 mg / 10 mg labeled
- Lot ID
- KC-RET-BLU1
- Tested
- Feb 17, 2026
Legal Notice
For research and laboratory use only. Retatrutide is an investigational compound that has not been approved for human use by any regulatory authority. Not for human consumption, in vivo use, or any medicinal application. Sold to qualified researchers and laboratories only. Customers must be 18 or older and are responsible for compliance with all applicable local laws and regulations.
Description
What is Retatrutide?
Retatrutide (laboratory designation LY3437943) is a synthetic peptide developed by Eli Lilly and Company. It is the first known unimolecular triple agonist that activates the receptors for glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon: three metabolic pathways that, in combination, are central to body weight regulation, glucose homeostasis, and energy expenditure. As of 2026, retatrutide is in Phase 3 clinical evaluation under the TRIUMPH trial program for chronic weight management and type 2 diabetes.
How it differs from existing GLP-1 drugs
Earlier-generation drugs in this class, semaglutide for example, are single GLP-1 agonists. Tirzepatide added a second mechanism (GIP). Retatrutide adds a third: glucagon receptor activation, which preclinical and Phase 1/2 work suggest contributes to increased lipolysis and energy expenditure rather than appetite suppression alone. The result observed in published Phase 2 data is the largest mean body-weight reduction reported for any peptide in this class to date.
/ Complete your protocol
What you'll also need
Everything for safe, sterile administration. Bundle in one order to save on shipping.